
An adaptive clinical trial of Evusheld and COVID-19 vaccination in immunosuppressed patients highly vulnerable to infection with COVID-19
What would my involvement be?
The full eligibility criteria for the study can be found via the following link:
https://www.isrctn.com/ISRCTN53507177
Eligible conditions include
Cohort 1: Haematological malignancies
Patients with a diagnosis of haematological malignancies and receiving one the following treatments:
a. Cohort 1a - Patients receiving active therapy with immunosuppressive or immunomodulating agents including:
- B-cell targeted therapies (rituximab or other anti-B-cell Ab therapy) used either as monotherapy or in combination with cytotoxic therapy in lymphoma / chronic lymphoproliferative disorders
OR
- JAK inhibitors (ruxolitinib or equivalent) in myeloproliferative neoplasms (MPNs)
OR
-
ImiD drugs (thalidomide, lenalidomide, pomalidomide or equivalent) in myeloma and other plasma cell dyscrasias
OR
- BTK inhibitors (ibrutinib or equivalent) in chronic lymphocytic leukaemia / chronic lymphoproliferative disorders
OR
- Treatment within 24 months with lymphodepleting agents followed by chimaeric antigen receptor (CAR) T-cell therapy for any haematological malignancy
b. Cohort 1b - Patients receiving aggressive therapy expected to cause temporary ablation of immune function including:
- Acute leukaemia (AML or ALL) being treated with curative intent using intensive combination chemotherapy schedules (excluding acute promyelocytic leukaemia)
OR
- Patients within 24 months of receipt of allogeneic stem cell transplant or receiving systemic immunosuppression for graft versus host disease.
-
Cohort 2: Solid tumours
- Early (Cohort 2a) or advanced cancer (solid organ) (Cohort 2b) undergoing systemic cancer treatment. Hormone treatments given alone such as tamoxifen or an aromatase inhibitor or LHRH agonist for early or advanced breast and prostate cancer are not considered to be a systemic treatment.
- Cancer patients are considered eligible for the trial if they have had a dose of systemic treatment within 42 days of trial
Cohort 3: Renal/hepatic disorders
Cohort 3a - Renal disorders:
Patients with kidney disease who fall into one of the following groups:
- Currently receiving immunosuppression
- Dialysis – including in-centre and home haemodialysis, peritoneal dialysis
- Transplant recipient receiving immunosuppression
Cohort 3b - Hepatic disorders
Patients with liver cirrhosis, autoimmune liver disease and liver transplant.
Cohort 4:Inflammatory disease.
- Patients receiving T-cell co-stimulation modulators, B-cell targeted therapies (including rituximab), tumour necrosis factor inhibitors (TNFi), soluble TNF receptors, interleukin (IL)-6 receptor inhibitors, IL-17 inhibitors, IL 12/23 inhibitors, IL 23 inhibitors or JAK inhibitors
- Patients who had received any immunotherapies listed above in the previous 3 months for autoimmune diseases, except in the case of rituximab treatment within the previous 6-month period.
- Patients receiving or had received in the previous 6 months immunosuppressive chemotherapy
- Patients receiving systemic immunosuppression for a chronic inflammatory disorder.
- Cohort 4a will include participants receiving rituximab and cohort 4b will include participants receiving non-rituximab immunosuppressants.
If you are eligible for the study, you will be asked to attend clinic to receive Evusheld. To take part in the study, you will need to:
- Provide written informed consent.
- Be able and willing to comply with all trial requirements.
- Be able and willing to practice continuous effective contraception during the first 3 months of the trial and, if appropriate, a negative pregnancy test on the day of screening.
- Provide access to all medical records with respect to current and past medical treatments.
- Have one or more of the following eligible conditions: haematological malignancy, solid tumour, renal disorder, hepatic disorder and/or inflammatory disorder.
- Be over 18 years old.
If you decide to take part in the study, you will be asked to sign the consent form and your General Practitioner will be notified of your participation. The treatment you are receiving for your current health condition will not be affected by your participation in the study and will continue as intended by your treating doctor. Your study doctor will monitor your safety and well-being throughout the study.
If you agree to take part in this study, you must come to the hospital at the times agreed with your study doctor as the information we will collect at these appointments are critical for answering the question of the study.
You will be asked to attend the clinic to provide blood samples 7 times over the space of one year to check the levels of Evusheld and to examine your immune responses against COVID-19. You will be compensated for your time.
Day 0
- Physical exam
- Pregnancy test (where applicable)
- Blood samples - before you receive Evusheld
- You may also be asked to provide a PCR swab for COVID-19 to see if you have a current infection.
- Receive Evusheld (600MG). This will be given as intramuscular injections. We will ask you to stay at the clinic for 15 minutes so that we can check on how you are. During this time, we will ask you to complete three questionnaires.
Day 14
Telephone call from your treating team who will ask you about how you are feeling and if you have had any side effects.
Day 28
- Blood sample
- Clinical assessment
- You may also be asked to provide a PCR swab for COVID-19 to see if you have a current infection.
- Receive your COVID-19 booster vaccine (Moderna vaccine or Pfizer/BioNTech vaccine). The type of vaccine booster you will receive will be randomly selected by a computer and will be administered through an injection in your arm. Your doctor will inform you which vaccine you have been allocated to as part of the study.
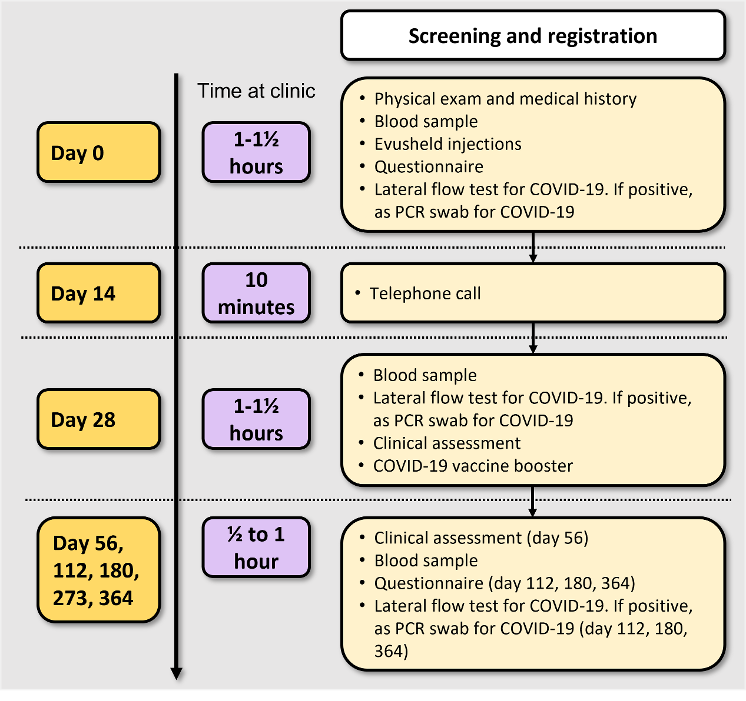
Follow-up visits
You will be asked to attend the clinic for follow up visits so that we can check how you are, to check on your symptoms and to provide further blood samples. These will be scheduled at56, 112, 180, 273 and 364 days post injection to check the levels of Evusheld and to examine your immune responses against COVID-19. At these visits, you may be asked to complete a questionnaire and a PCR swab for COVID-19.
If you become symptomatic for COVID-19 during the study you must take a lateral flow test and self-report the result to the study team. In this eventuality, you will be asked questions about the severity of your symptoms and will be required to take a nasal swab at home, and return this in the freepost packaging to the laboratory for PCR testing. The materials and instructions for collecting, packaging and posting swabs will be provided to you as part of the study.
You are encouraged to keep a supply of lateral flow tests at home whilst you are on the study. We advise you to regularly order via the NHS website: www.gov.uk/order-coronavirus-rapid-lateral-flow-tests
What would the questionnaires involve?
You will be asked to complete questionnaires at four timepoints throughout the study (Days 0, 112, 180 and 364 days after the Evusheld injection). The questionnaire should take about 10-15 minutes to complete.
The questionnaires ask about your household, behaviour changes due to the COVID-19 pandemic and your general health and wellbeing. Your unique trial number will be collected, in addition to your partial date of birth (mm/yyyy), when completing the questionnaires to link your trial data. The collection of partial date of birth on these questionnaires is as an extra cross check to ensure that the correct data is being collected for the correct participant and will not be used for any other reason.
